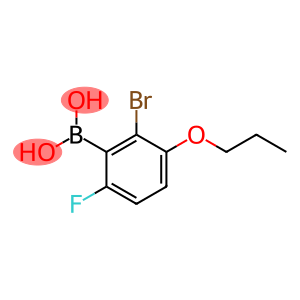
Boronic acid, B-(2-bromo-6-fluoro-3-propoxyphenyl)-
(2-bromo-6-fluoro-3-propoxyphenyl)boronic acid
CAS: 849052-20-6
Molecular Formula: C9H11BBrFO3
Boronic acid, B-(2-bromo-6-fluoro-3-propoxyphenyl)- - Names and Identifiers
| Name | (2-bromo-6-fluoro-3-propoxyphenyl)boronic acid |
| Synonyms | 2-Bromo-6-fluoro-3-proproxyphenylboronic acid (2-bromo-6-fluoro-3-propoxyphenyl)boronic acid Boronic acid, B-(2-bromo-6-fluoro-3-propoxyphenyl)- |
| CAS | 849052-20-6 |
| InChI | InChI=1/C9H11BBrFO3/c1-2-5-15-7-4-3-6(12)8(9(7)11)10(13)14/h3-4,13-14H,2,5H2,1H3 |
Boronic acid, B-(2-bromo-6-fluoro-3-propoxyphenyl)- - Physico-chemical Properties
| Molecular Formula | C9H11BBrFO3 |
| Molar Mass | 276.9 |
| Density | 1.53g/cm3 |
| Melting Point | 83-88°C(lit.) |
| Boling Point | 394.1°C at 760 mmHg |
| Flash Point | 192.1°C |
| Vapor Presure | 6.43E-07mmHg at 25°C |
| Storage Condition | 2-8°C |
| Refractive Index | 1.542 |
Boronic acid, B-(2-bromo-6-fluoro-3-propoxyphenyl)- - Risk and Safety
| WGK Germany | 3 |
Boronic acid, B-(2-bromo-6-fluoro-3-propoxyphenyl)- - Introduction
boronic acid is an organic compound with the chemical formula C9H10BBrFNO3.
The properties of the compound are as follows:
-Appearance: colorless or yellowish crystal powder
-Melting point: about 150-155°C
-Solubility: Slightly soluble in water, soluble in organic solvents such as dimethyl sulfoxide (DMSO), methanol and dichloromethane
(br) boronic acid is mainly used in the field of organic synthesis. It is commonly used as a reagent for aryl boronic acid to participate in organic reactions, such as palladium-catalyzed boron alcohol esterification reaction, Suuki coupling reaction, Stille coupling reaction, etc. It can be used as an important aryl boronic acid reagent or intermediate in organic synthesis for the construction of complex organic molecules.
(1) The preparation method of boronic acid is generally obtained by the substitution reaction of the compound. A specific step may be to sequentially substitute bromine, fluorine and propoxy.
For safety information, due to limited toxicological and safety data boronic acid, it is recommended to follow laboratory safety practices and wear appropriate personal protective equipment, such as lab gloves, goggles and lab coats. At the same time, avoid contact with skin and eyes, avoid breathing dust or steam. In case of accidental contact, rinse immediately with plenty of water and consult a doctor. In addition, the compound should be stored in a cool, dry place, away from fire and oxidizing agents.
The properties of the compound are as follows:
-Appearance: colorless or yellowish crystal powder
-Melting point: about 150-155°C
-Solubility: Slightly soluble in water, soluble in organic solvents such as dimethyl sulfoxide (DMSO), methanol and dichloromethane
(br) boronic acid is mainly used in the field of organic synthesis. It is commonly used as a reagent for aryl boronic acid to participate in organic reactions, such as palladium-catalyzed boron alcohol esterification reaction, Suuki coupling reaction, Stille coupling reaction, etc. It can be used as an important aryl boronic acid reagent or intermediate in organic synthesis for the construction of complex organic molecules.
(1) The preparation method of boronic acid is generally obtained by the substitution reaction of the compound. A specific step may be to sequentially substitute bromine, fluorine and propoxy.
For safety information, due to limited toxicological and safety data boronic acid, it is recommended to follow laboratory safety practices and wear appropriate personal protective equipment, such as lab gloves, goggles and lab coats. At the same time, avoid contact with skin and eyes, avoid breathing dust or steam. In case of accidental contact, rinse immediately with plenty of water and consult a doctor. In addition, the compound should be stored in a cool, dry place, away from fire and oxidizing agents.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: 2-Bromo-6-fluoro-3-propoxyphenylboronic acid Request for quotation
CAS: 849052-20-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 849052-20-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 3-BROMO-6-FLUORO-3-PROPOXYPHENYLBORONIC& Visit Supplier Webpage Request for quotation
CAS: 849052-20-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 849052-20-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 2-Bromo-6-fluoro-3-propoxyphenylboronic acid Request for quotation
CAS: 849052-20-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 849052-20-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 3-BROMO-6-FLUORO-3-PROPOXYPHENYLBORONIC& Visit Supplier Webpage Request for quotation
CAS: 849052-20-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 849052-20-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

